|
But be patient as posts will appear after passing our moderation. Depending on the number of messages we receive, you could wait up to 24 hours for your message to appear. And thus the abundance of boron-11 is roughly 81. Where u is the unit for atomic mass and x is the proportion of boron-10 out of the total boron abundance which is 100. Questions will be queued for posting immediately after moderationģ. And 10.81 u is a lot closer to 11u than it is to 10u, so there must be more of boron-11. Answers to questions will be posted immediately after moderationĢ. Read the community guidelines MathsGee Rulesġ. For example, the element boron is composed of two isotopes: About 19.9 of all boron atoms are 10 B with a mass of 10.0129 amu, and the remaining 80.1 are 11 B with a mass of 11.0093 amu. 0.24 compound contains 0.096 g of Boron 100 g compound contains 0.096. Join expert live video sessions (Paid/Free)Ĥ. Concentration of solution 100 20 5 Mass of solute ×100 (Mass of solute +. Boron-12 B CID 58665377 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. We use a combination of generative AI and human experts to provide you the best answers to your questions. The atomic mass of one isotope is 11, the mass of other isotope is: A 10.81 B 11.01 C 10.05 D 21.82 Medium Solution Verified by Toppr Correct option is C) Atomic mass of Boron 10. Then the mass number is total protons plus neutrons. It has two isotopes with 80 and 20 abundance respectively. Look up properties, history, uses, and more. So boron-11 has five protons the same as boron-10. Chemical element, Boron, information from authoritative sources. One atomic mass unit is equal to 1.66 x 10 -24 grams.  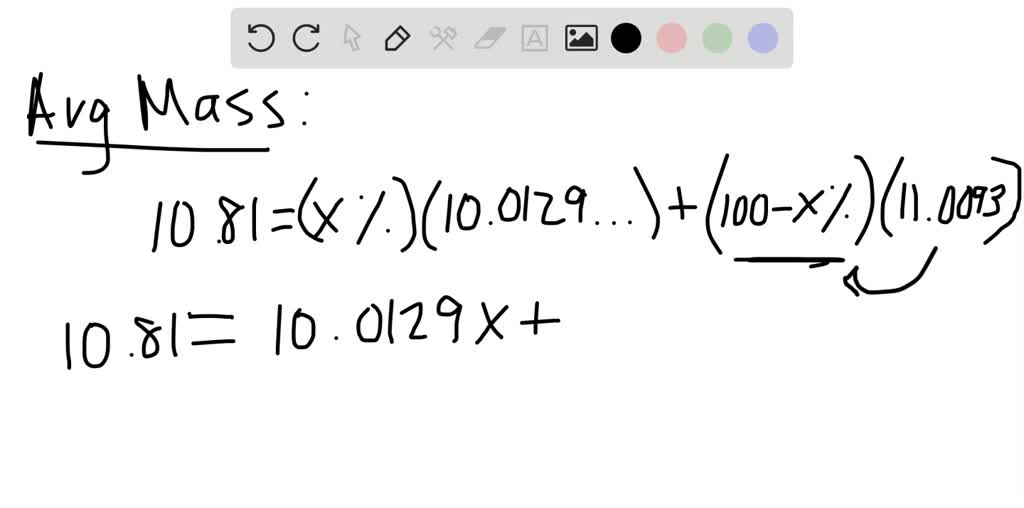
The unit of measure for mass is the atomic mass unit (amu). Therefore this resulting atomic mass is calculated from naturally-occurring isotopes and their abundance. Boron is a nonmetallic element, which has five protons in its nucleus along with five to six neutrons, resulting in two stable isotope forms: 10B (abundance. We are given that boron-10 had five protons in its nucleus, and any element always has the same number of protons in its nucleus (atomic number). Note that each element may contain more isotopes.  
If you are ever confused, remember that the atomic number should always be the smaller of the two and will be a whole number, while the atomic mass should always be the larger of the two and will be a decimal number.Join MathsGee, where you get instant answers from our AI, GaussTheBot, that are verified by human experts. Isotopes are given by the name of the element and the mass number. Take time to notice that not all periodic tables have the atomic number above the element's symbol and the mass number below it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
